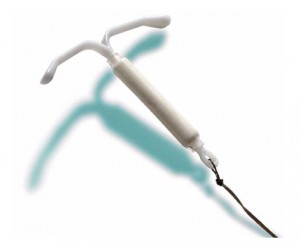
Have you been seriously injured by the Mirena IUD? Mirena IUD lawsuits are being filed by women around the country who allege the intrauterine device caused serious side effects, injuries, including uterine perforations. According to the Mirena lawsuits, the IUD can migrate within a woman’s body, even outside of her uterus, and potentially cause a number of serious side effects and injuries, including:
- Abscesses
- Embedment in the uterine wall
- Uterine perforations
- Infertility
- Infection
- Intestinal perforations or obstruction
- Pelvic Inflammatory Disease
- Ectopic Pregnancy
Mirena is a small, t-shaped, plastic device that is inserted into the uterus by a healthcare provider and works to prevent pregnancy by releasing a low dose of levonorgestrel (a synthetic progestin hormone) directly into the uterus. Once a doctor inserts the Mirena IUD, it can stay in place for up to five years. Mirena was approved as a contraceptive by the U.S. Food & Drug Administration (FDA) in 2000, and is recommended for women who have already had one child. In 2009, the FDA expanded Mirena’s approval to treat heavy menstrual bleeding in women who prefer an intrauterine device for contraception.
A number of Mirena IUD lawsuits have been filed by woman who allege the device migrated from its position in the uterus, leading to perforation or puncturing of the uterus, migration of the device to other parts of the body, and infections that may diminish fertility or the ability to conceive. When the Mirena IUD migrates from its original placement, it will no longer work to prevent pregnancy. Women who become pregnant while using Mirena are more likely to experience an ectopic pregnancy, which occurs in the fallopian tube.
When a Mirena IUD migrates, a doctor must locate and surgically remove the device. If the device caused uterine punctures or perforations, abscesses, scarring or infection, further surgical intervention – including a hysterectomy in the most severe cases – may be required.
According to Mirena IUD lawsuits, Bayer, the manufacturer of the device, aggressively marketed Mirena to a “Busy Mom” demographic as a hassle-free form of birth control. In 2009, the FDA issued a warning letter to Bayer, after finding its Mirena promotions overstated the efficacy of the device, presented unsubstantiated claims, minimized the risks of Mirena, and used false and misleading presentations during in-home events touting the IUD. Among other things, the FDA took issue with marketing claims touting Mirena’s purported ability to improve a woman’s sex life and help her “look and feel great.”
If you or a woman you love were injured by Mirena, call the attorneys at Sabatini and Associates, LLC today at 860-667-0839.