 Legal claims are being brought by injured women who have undergone surgery to implant transvaginal mesh and pelvic/bladder support medical devices.
Legal claims are being brought by injured women who have undergone surgery to implant transvaginal mesh and pelvic/bladder support medical devices.
Transvaginal mesh and pelvic/bladder support medical devices are used to treat stress urinary incontinence (SUI) and pelvic organ prolapse (POP). These two conditions result in the weakening of of the pelvic region – a common problem after a hysterectomy or childbirth.
On July 13, 2011, the FDA released a medical alert after receiving reports of over 1500 cases where the use of transvaginal mesh and pelvic/bladder support devices caused serious injury to women. The FDA warned physicians that: (1) in most instances the risk of serious injury outweighed all purported benefits associated with the medical devices; (2) traditional procedures that do not use polypropylene mesh devices should be the primary course of treatment; and (3) the implantation of vaginal mesh/pelvic devices should be limited to last resort treatment cases. The reported injuries that these devices are inflicting upon women include:
1. Erosion of mesh into the vagina
2. Serious infection
3. Urinary problems
4. Pelvic pain
5. Vaginal pain
6. Hardening of the mesh
7. Injury to nearby organs
8. Painful intercourse
9. Recurrence of pelvic organ prolapse and stress urinary incontinence
10. Perforations created by the mesh in the bowel, bladder and/or blood vessels
11. Vaginal scarring and other complications
A recent clinical study was conducted to determine the safety of these products. The study was stopped because the problems being seen in the study in patients were too severe. More than 15% of the women in the study experienced the dangerous and painful condition known as “erosion” of the vaginal tissue in which the skins splits and the mesh protrudes. Sabatini and Associates, LLC is actively investigating legal claims on behalf of women who are alleging serious injuries associated with vaginal mesh and the firm is currently accepting new clients involving transvaginal mesh and pelvic/bladder support products.
Read More
In August 2010, the DePuy ASR metal-on-metal artificial hip device was recalled due to an increased rate of revisions (follow up surgeries) occurring shortly after the original hip implant. Certain medical literature and data has shown that a number of revisions in users of the ASR hip are due to metal debris from the metal components in the device. Following the ASR recall, a MDL was formed for all lawsuits filed in federal courts around the nation. 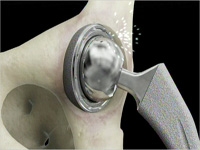
Now a second MDL has formed because DePuy makes various hip devices besides the ASR. Specifically, DePuy makes the Pinnacle metal-on-metal hip model that is similar to the ASR. There have been reports of metal poisoning and pre-mature revisions occurring with the Pinnacle device. Multiple lawsuits have been filed and the DePuy Pinnacle MDL has been assigned to Judge James E. Kinkeade in the Northern District of Texas.
Read More
The F.D.A. is conducting a review of certain birth control bills that contain the hormone drospirenone. The hormone is found in Bayer’s Yaz, Yasmin, Beyaz and Safyral. While all birth control pills pose a rik of blood clots, certain studies have suggested that Bayer’s birth control pills pose a greater risk of blood clots. Two recent reports in the British Medical Journal found a two-fold to threefold greater risk of blood clots in women taking birth control pills like Bayer’s Yaz. The F.D.A. ‘s current review involves an 800,000 person study. The sale of Yaz family of products in 2010 represented 3.3 percent of Bayer’s annual revenue. The lawyers at Sabatini & Associates, LLC currently represent women injured by Yaz and Yasmin and is accepting new cases.
Read More
Bailed out by the taxpayers, but off the hook for compensating some of the very same people who have been seriously injured by their defective motor vehicles, this is the deal Chrysler and GM got as part of their government bailouts. When the U.S. Government brokered a bankruptcy for Chrysler two years ago, it allowed the car maker to discharge any and all obligations it owed to car accident victims with pending cases against the automaker, or those who had already won an award or settlement. The Wall Street Journal has tracked several stories of some of the losers in the government deal, including the family of Vicki Denton. Ms. Denton died when the airbag in her 1998 Dodge Caravan failed to deploy in a collision. After years of litigation, in 2009 a jury determined that Ms. Denton’s vehicle was defective, and order Chrysler to pay her son $2.2 million in damages. Despite the jury’s finding, Chrysler has not paid the judgment, and under the rules of the bailout will never have to.
Read More
Realizing that they had a major problem with the defective ASR hip implant, J&J and DePuy established a “medical bill reimbursement program”. The program allowed DePuy to pay for the medical bills related to the medical treatment care and treatment necessitated by the defective hip implants including revision surgeries. However, for a patient with a defective hip implant to access the program, the patient was being required to sign and hand over a medical authorization to DePuy. Lawyers representing clients have put an end to this requirement. Clients can now access the medical reimbursement program without signing the medical authorization. All information related to the client’s case goes through the lawyer representing the client. This ensures that the client’s rights are well protected. This also means that for injured ASR hip users without current legal representation, it is important that the person retain legal counsel. For more information or to inquire about retaining legal representation, feel free to contact Attorney James Sabatini, the firm’s lawyer handling hip implant litigation.
Read More
 Legal claims are being brought by injured women who have undergone surgery to implant transvaginal mesh and pelvic/bladder support medical devices.
Legal claims are being brought by injured women who have undergone surgery to implant transvaginal mesh and pelvic/bladder support medical devices.


